A zebra finch can fit in the palm of your hand, but its brain is doing something that looks almost unruly.
Inside one part of the adult songbird brain, newly formed neurons do not politely weave around older cells as they settle into place. Instead, they appear to push through crowded tissue and press into neighboring neurons. They bend nearby structures and, at times, seem to carve tunnels through tightly packed cell groups. That unexpected behavior, described by researchers at Boston University, offers a striking new look at how adult brains in some animals keep adding neurons long after birth.
The work centers on neurogenesis, the process by which neurons are born, migrate, mature, and join existing brain circuits. In most mammals, that ability is sharply limited after birth. Birds, fish, and reptiles are different. Their brains continue to refresh themselves, and zebra finches are especially good at it.
That makes them valuable for studying a basic puzzle. If some animals can keep adding neurons to adult brains, how do those cells actually move through tissue that is already crowded with mature cells, stable synapses, and long-established connections tied to behavior and memory?
Benjamin Scott, an assistant professor of psychological and brain sciences at Boston University and the study’s corresponding author, said the team expected new neurons to skirt around existing structures. What they saw instead was far more forceful.
“We found that in songbirds, new neurons in the adult brain behave like explorers forging a path through a dense jungle,” Scott said.
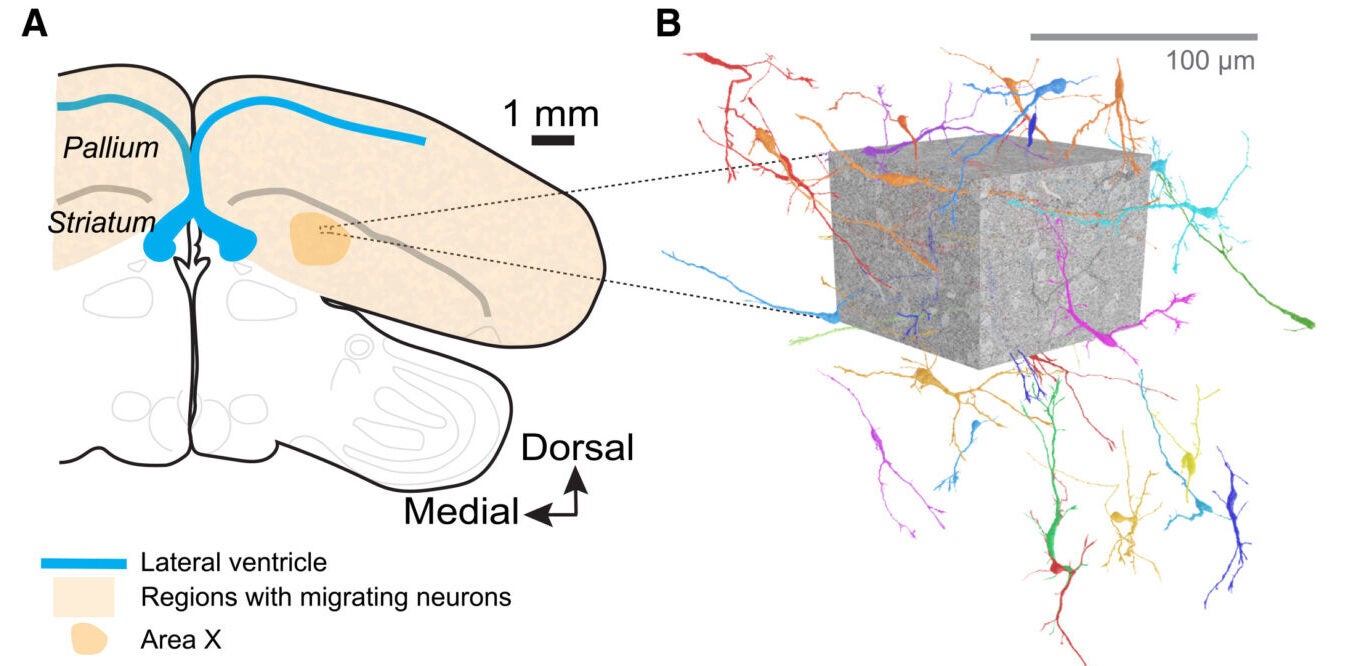
The team focused on zebra finches because they are known as strong models for adult neurogenesis. New neurons are added to parts of the song system, including regions involved in learning and producing song. In this study, the researchers examined Area X, a region in the striatum linked to song learning.
To do that, they turned to electron microscopy-based connectomics, a form of imaging that can capture brain tissue at extremely high resolution. Scott said the original goal was simple: get a close look at what this process physically resembles in adult brain tissue.
The dataset came from the first fully reconstructed zebra finch connectome from Area X. The researchers identified 35 migratory neurons with largely complete reconstructions for detailed analysis. They did this using fluorescence microscopy and a machine-learning classifier trained on known cell shapes. These cells had features expected of immature, moving neurons. For instance, they had elongated cell bodies, a leading process, finger-like extensions, and clustered mitochondria near the centrosome.
From there, the surprise grew. The migratory neurons were not tucked into empty corridors. Instead, they were scattered widely through dense brain tissue and oriented in multiple directions. Their density was estimated at 1,390 neurons per cubic millimeter in the sampled volume. Moreover, their distribution fit a model consistent with broad, uniform dispersal.
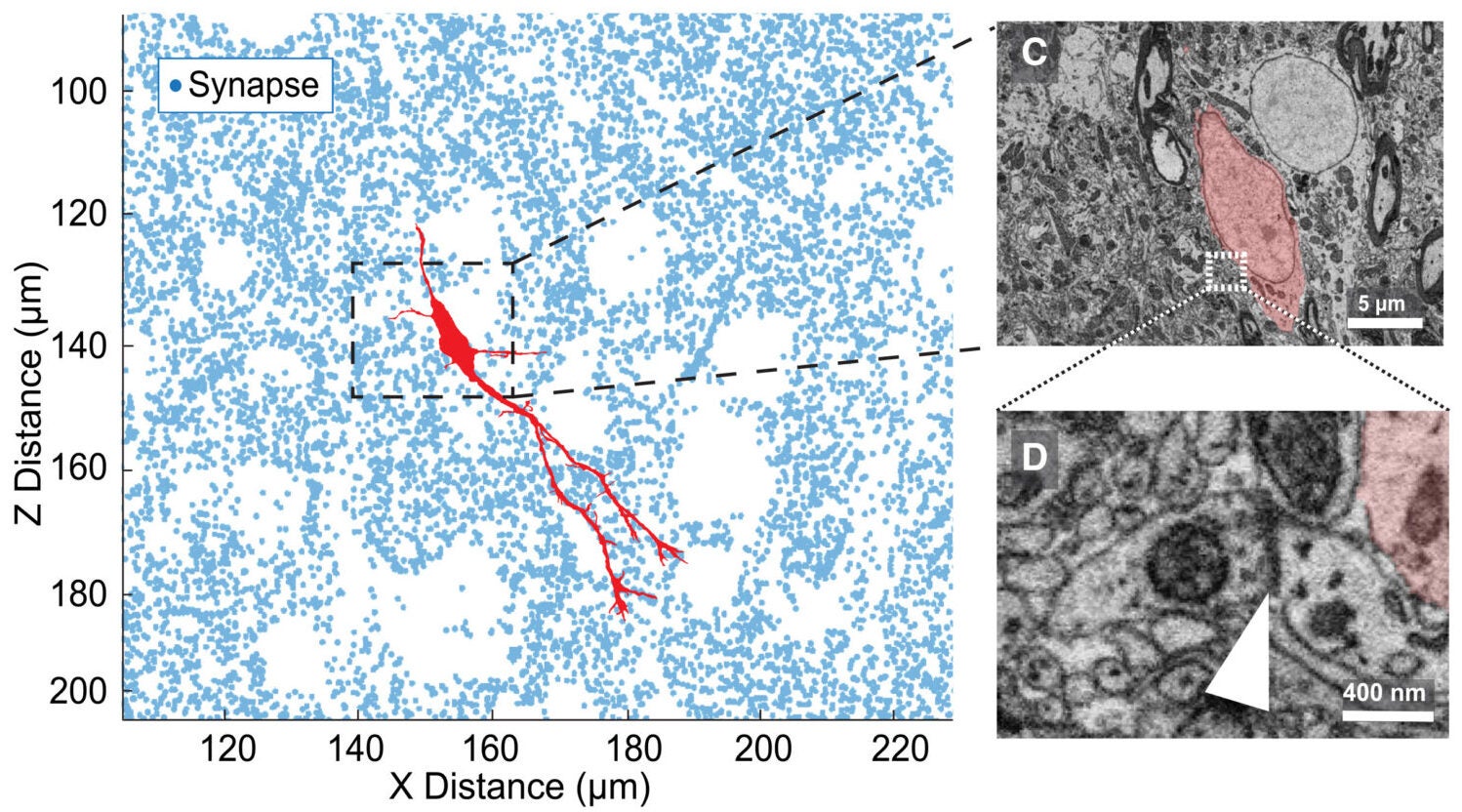
The surrounding environment was anything but open. The connectome contained more than 8 million high-confidence synapses, with a density of 0.29 per cubic micrometer. Yet the local synapse density around the migratory neurons was not significantly different from the broader tissue around them.
In other words, these new cells were moving through a synapse-rich thicket.
The researchers found that migrating neurons made frequent contact with the mature structures around them, including axons, dendrites, and neuronal spines. In some places, nearby dendrites curved around a migratory cell body. Synapse density even rose immediately around the soma of these new neurons, suggesting that the surrounding tissue may be getting compressed rather than avoided.
Only a small minority of the 35 migratory neurons, 8 cells, were associated with known scaffolds such as radial fibers or blood vessels, structures that often help guide migrating cells in other systems.
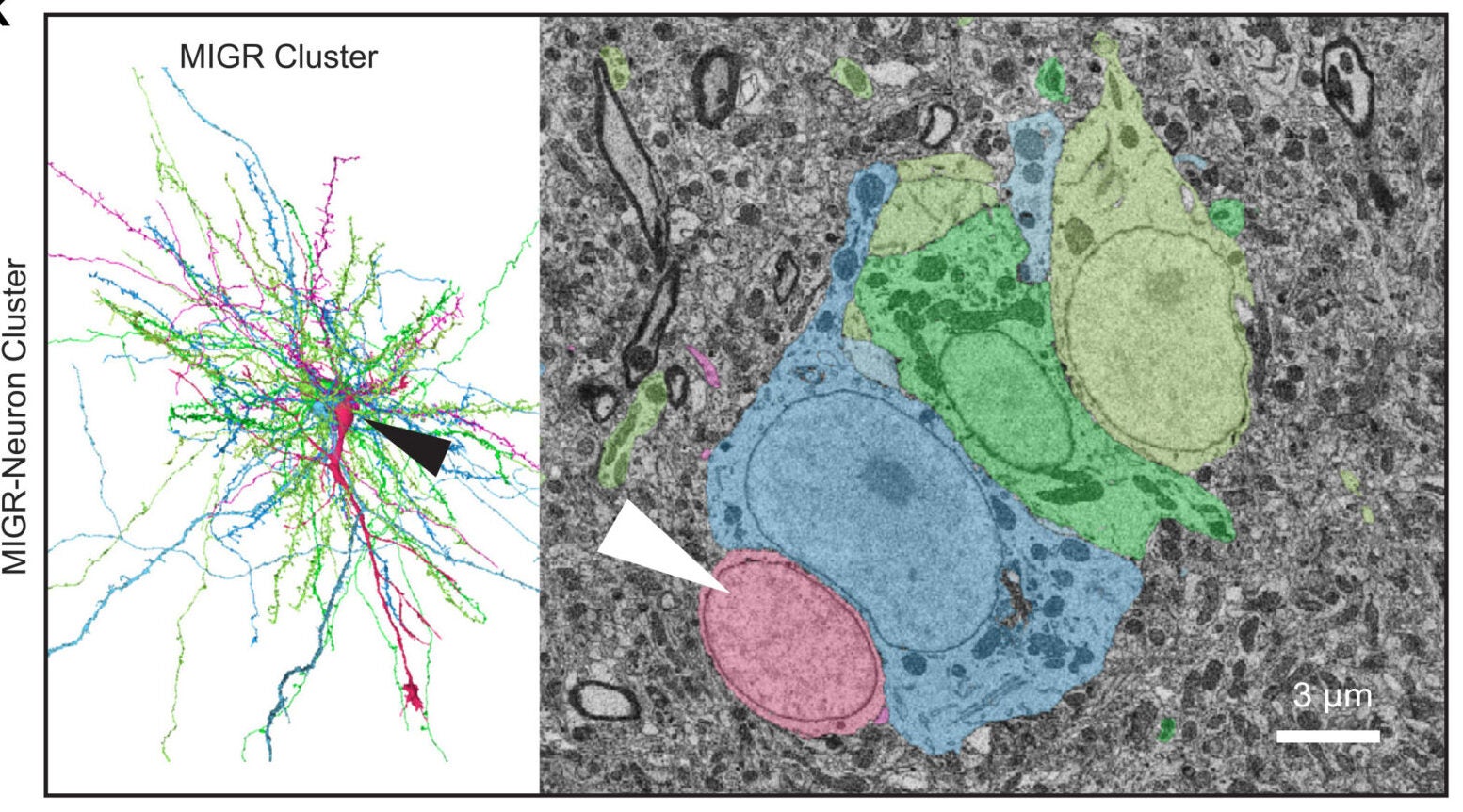
Instead, close contact with mature neurons was common. Seventeen of the 35 migratory neurons formed soma-to-soma associations with mature neurons. Four also associated with astrocytes.
When the researchers measured those points of contact, they found the interactions were not balanced. Mature neuron bodies were deformed far more than the migratory neurons themselves. The average indentation depth in mature neurons was 2.03 micrometers, compared with 0.59 micrometers in the migratory cells. In several cases, the new neurons sat inside large concavities involving one or more mature neurons.
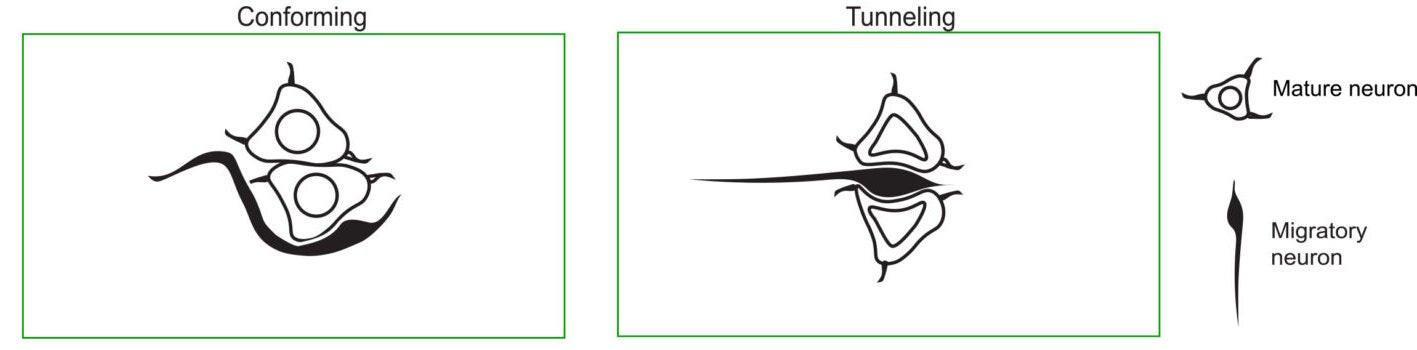
That is where the study’s most vivid term enters: tunneling.
The researchers used it to describe cases in which a migratory neuron appeared to deform several neighboring cell bodies, axons, and dendrites as it moved through densely packed tissue. In some examples, the new neuron seemed to pass through or into clusters of mature neurons rather than around them.
This kind of neuronal tunneling has not, to the researchers’ knowledge, been described before in the vertebrate nervous system.
The finding raises a harder question than simple cell movement. If new neurons physically disturb established tissue, what does that cost the brain?
Scott said one possibility is that limiting neurogenesis after birth may help protect the human brain. Mature mammalian brains rely on stable connections to preserve memory and function. A cell that barges through that environment could damage stored information or disrupt circuits.
“This potentially disruptive behavior may help explain why humans and other mammals have limited capacity to regenerate brain tissue in adulthood,” Scott said, “leaving us more vulnerable to neurodegenerative disorders such as Alzheimer’s disease.”
He also offered a more hopeful interpretation. Most glial scaffolds, the cellular highways that help guide migrating neurons, disappear in humans after birth. That loss has been seen as one obstacle to adult neurogenesis. But in the zebra finch, the new neurons in this study often moved without those scaffolds.
“Our discovery of tunneling shows how cells can move without glia scaffolds,” Scott said.
If that principle carries beyond birds, it could matter for future brain repair strategies, including stem cell approaches aimed at generating or guiding new neurons in adult human brains.
The work also draws an intriguing parallel outside neuroscience. The researchers noted that tunneling-like behavior has been described in metastatic cancer cells moving through confined tissue.
Still, the authors were careful about the limits of their findings.
Chemical fixation and electron microscopy processing can alter tissue dimensions and reduce extracellular space, which may affect how cramped the tissue appears. The dataset’s voxel resolution also limited the team’s ability to see very fine structures. Furthermore, annotation or segmentation errors could affect interpretation. The imaged brain block covered only a restricted region of Area X, several millimeters from where these neurons are born. That means the study may underrepresent other forms of migration, including glial-dependent routes seen elsewhere in the avian brain.
The researchers also noted that tunneling may mark a later stage of migration, rather than the whole journey.
This study gives scientists a more concrete picture of how adult-born neurons can move through an already functioning brain. It suggests that in species with lifelong neurogenesis, adding new neurons may involve physical disruption as well as renewal.
That matters for efforts to understand memory, learning, and brain repair. It also opens a new line of research into whether adult brain regeneration requires special scaffolds, or whether cells can be guided through dense tissue in other ways.
For medicine, that could shape how researchers think about stem-cell therapies, injury recovery, and why the human brain has such limited capacity to replace lost neurons.
Research findings are available online in the journal Current Biology.
The original story “Zebra finch neurons offer new clues about learning, repair, and human brain limits” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post Zebra finch neurons offer new clues about learning, repair, and human brain limits appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.